Double Cross-Linked Polyisobutylene-Based Polyurethane (xPIB-PU) I: Synthesis, Processing, and Properties
Turgut Nugay*, Elif Kurnaz, Sina Naficy, Joseph P. Kennedy*
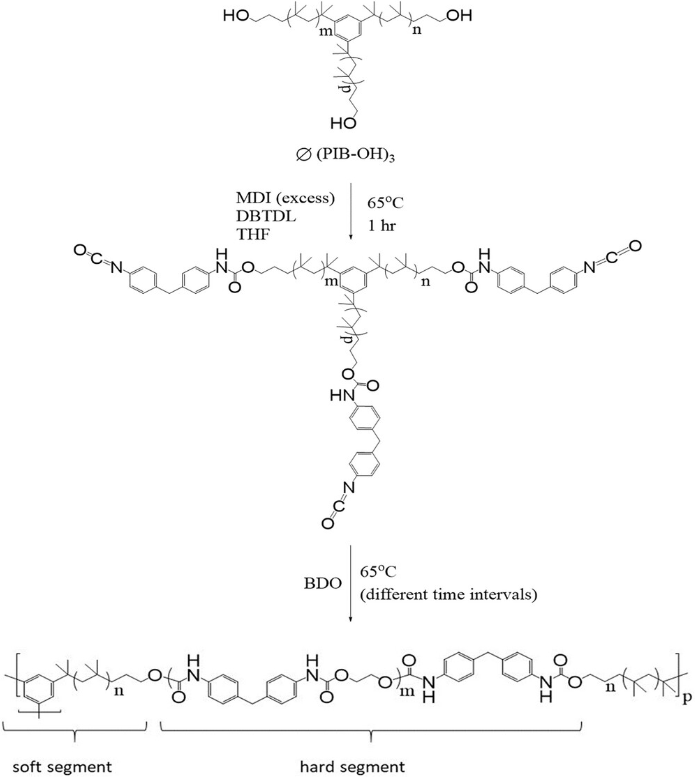
This publication concerns the synthesis of a novel class of hybrid thermoset/thermoplastic elastomers, i.e., double cross-linked polyisobutylene-based polyurethanes (xPIB-PUs), together with their processing and some key properties. These materials are designed to exhibit a combination of properties for indwelling medical applications, specifically for heart valve prostheses. The chemical cross-links are introduced by well-defined 3-arm PIB stars fitted with HO-end groups of molecular weights (MWs) of Mn = 3081 and 9000 g mole−1 and narrow MW dispersities (<1.3). Processibility of xPIB-PU containing 70 wt% PIB (for biocompatibility and calcification resistance) is achieved by driving the syntheses to high conversions (>97%) close to but still below the gel point and completing the cross-linking in heated molds. The processibility period (minutes) is controlled by controlling the MW of the cross-linking agent in the 3081 to 9000 g mole−1 range. Blends of 3-arm stars and linear bifunctional PIBs can also control the processibility period. The creep resistance of xPIB-PU is significantly improved (from 13.2% to 5.5%) relative to physically cross-linked PIB-PU. xPIB-PUs exhibit negligible permanent set (1–2%), and high Young’s modulus (41 MPa). The stress/strain properties of xPIB-PUs are also investigated. The data clearly show the contribution of two modes of cross-linking.
Macromolecular Chemistry and Physics 2024, 225(11), 2300406
Structural reinforcements as a strategy toward durable polymeric heart valves
Jacopo Giaretta, Matthew Crago, Thanh Phuong Hoang, Xinying Liu, Aeryne Lee, Yiqi Wang, Syamak Farajikhah, David Frederick Fletcher, Fariba Dehghani, David Scott Winlaw, Sina Naficy*
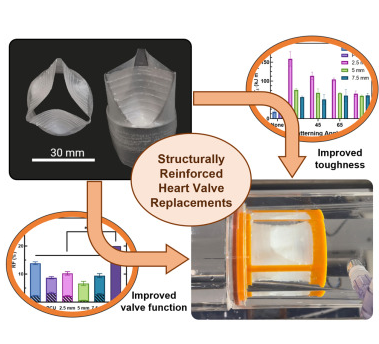
Valvular heart diseases are commonly addressed by the implantation of heart valve replacement devices; however, current prosthetics remain suboptimal in their performance and longevity, often necessitating multiple operations. Here, structurally reinforced polymeric heart valves are fabricated, utilizing a combination of dip coating and 4-axis three-dimensional printing with different grades of elastomeric polyurethanes. This strategy introduces mechanical anisotropy, increases toughness nearly 9-fold, and enhances fatigue resistance with a 150% increase in the energy required for tear propagation. While all valves exceed the ISO 5840-2 standard, structural reinforcement prevents the progression of tears during accelerated cardiac cycling and reduces regurgitation by about 50% compared with unreinforced valves without compromising opening performance. Subsequently, we demonstrate a valved conduit to broaden the possible clinical applications of this design. These structurally reinforced polymeric heart valve replacements could offer improved device performance and resistance against tear propagation, thus providing a vital window for necessary clinical interventions.
Cell Reports Physical Reports 2024, 5(3), 101870
Protein adsorption on blood-contacting surfaces: A thermodynamic perspective to guide the design of antithrombogenic polymer coatings
Matthew Crago, Aeryne Lee, Thanh Phuong Hoang, Sepehr Talebian*, Sina Naficy*
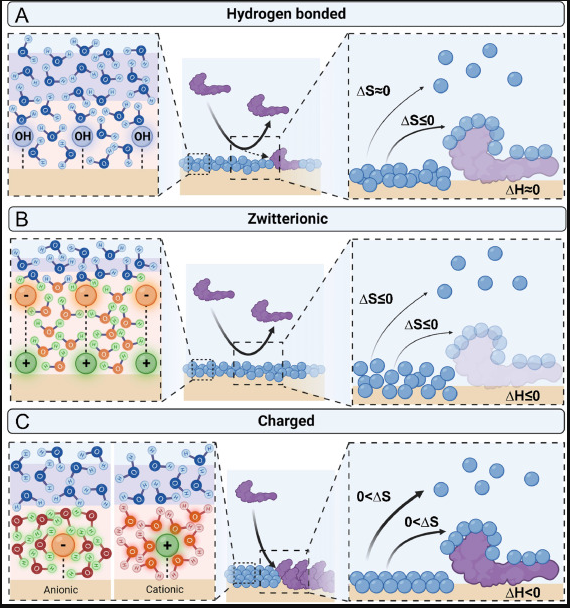
Blood-contacting medical devices often succumb to thrombosis, limiting their durability and safety in clinical applications. A plethora of polymeric coating modifications have been utilized for addressing this issue. This review offers a cohesive understanding of the thermodynamics governing these phenomena, to specifically aid in the design and selection of hemocompatible polymeric coatings for biomedical applications.
Acta Biomaterialia 2024, 108, 46-60

Durable plasma-mediated zwitterionic grafting on polymeric surfaces for implantable medical devices
Matthew Crago, Richard Tan, Juichien Hung, Steven G. Wise, Behnam Akhavan, Marcela Bilek, Fariba Dehghani, Sepehr Talebian, Sina Naficy*
Adverse body reactions to blood-contacting medical devices endanger patient safety and impair device functionality, with events invariably linked to nonspecific protein adsorption due to suboptimal material biocompatibility. To improve the safety and durability of such devices, herein we propose a strategy for introducing stable zwitterionic grafts onto polymeric surfaces via plasma functionalization.
The resulting zwitterion-grafted substrates exhibit long-lasting superhydrophilicity, enabling antifouling and anti-thrombogenic properties. We demonstrate the successful modification of the surface elemental composition, morphology, and hydrophilicity, while retaining the underlying mechanical properties of the polymeric substrate. Furthermore, we optimise the fabrication process to ensure long-lasting modifications at least three months after fabrication. This strategy decreases fibrinogen adsorption by approximately 9-fold, and thrombosis by almost 75% when applied to a commercial polyurethane. Moreover, this process is universally applicable to a wide range of polymeric materials, even those with stable chemistry such as polytetrafluoroethylene.
Communications Materials 2024
Figure. Water builds up on the superhydrophilic surface after treatment with the disclosed method.
Bioinspired Polymeric Heart Valves: A Combined In Vitro and In Silico Approach
Aeryne Lee, Xinying Liu, Jacopo Emilio Giaretta, Thanh Phuong Hoang, Matthew Crago, Syamak Farajikhah, Luke Mosse, David Frederick Fletcher, Fariba Dehghani, David Scott Winlaw, Sina Naficy*
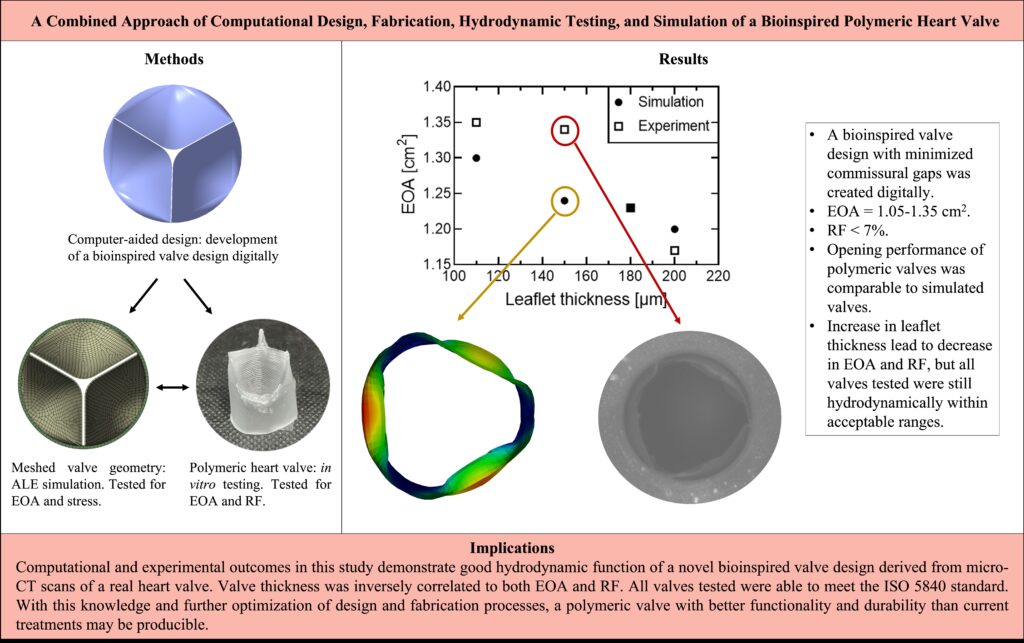
Polymeric heart valves (PHVs) may address the limitations of mechanical and tissue valves in the treatment of valvular heart disease. In this study, a bioinspired valve was designed, assessed in silico, and validated by an in vitro model to develop a valve with optimum function for pediatric applications.
A bioinspired heart valve was created computationally with leaflet curvature derived from native valve anatomies. A valve diameter of 18 mm was chosen to approach sizes suitable for younger patients. Valves of different thickness were fabricated via dip-coating with siloxane-based polyurethane and tested in a pulse duplicator for their hydrodynamic function. The same valves were tested computationally using an Arbitrary Lagrangian-Eulerian (ALE) plus immersed solid approach, where the fluid structure interaction between the valves and fluid passing through them was studied and compared with experimental data.
Computational analysis showed valves of 110-200 μm thickness had effective orifice areas (EOAs) of 1.20-1.30 cm2 with thinner valves exhibiting larger openings. In vitro tests demonstrated PHVs of similar thickness had EOAs of 1.05-1.35 cm2 and regurgitant fractions (RFs) less than 7%. Valves with thinner leaflets exhibited optimal systolic performance while thicker valves showed lower RFs.
Bioinspired PHVs demonstrated good hydrodynamic performance that exceeded ISO 5840-2 standards. Both methods of analysis showed similar correlations between leaflet thickness and valve systolic function. Further development of this PHV may lead to enhanced durability and therefore become a more reliable heart valve replacement than contemporary options.
The Journal of Thoracic and Cardiovascular Surgery Open 2023, 15, 113-124
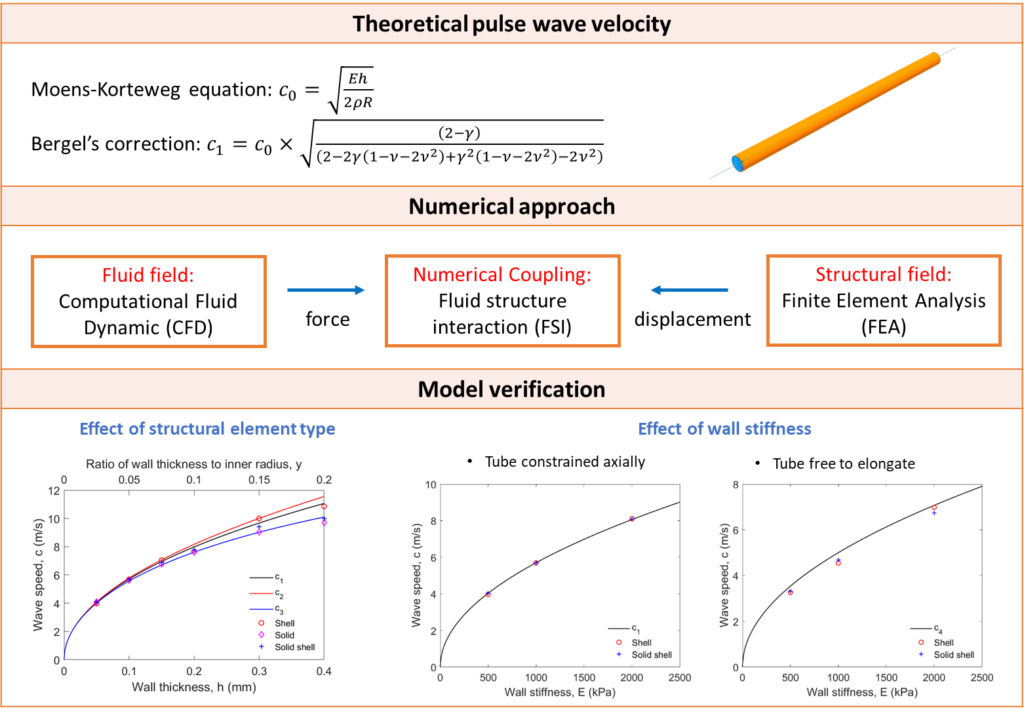
Verification of fluid-structure interaction modelling for wave propagation in fluid-filled elastic tubes
Xinying Liu, David F. Fletcher*
This paper presents a verification study of wave propagation in fluid-filled elastic tubes using a coupled numerical simulation method by comparing the simulation results with analytical solutions. A three-dimensional fluid-structure interaction numerical model is built using Ansys software. Wave propagation is investigated by applying a pressure pulse at the inlet of a fluid-filled elastic tube. The speed of the pressure wave and the radial displacement of the tube are simulated and compared with theoretical values. Simulation results yield a high level of accuracy. Different structural elements are used to represent the tube, and their impact on the results is discussed. The effects of tube material, tube constraints and fluid properties are also investigated in this study.
Journal of Algorithms & Computational Technology 2023, 17, DOI: 10.1177/17483026231159793
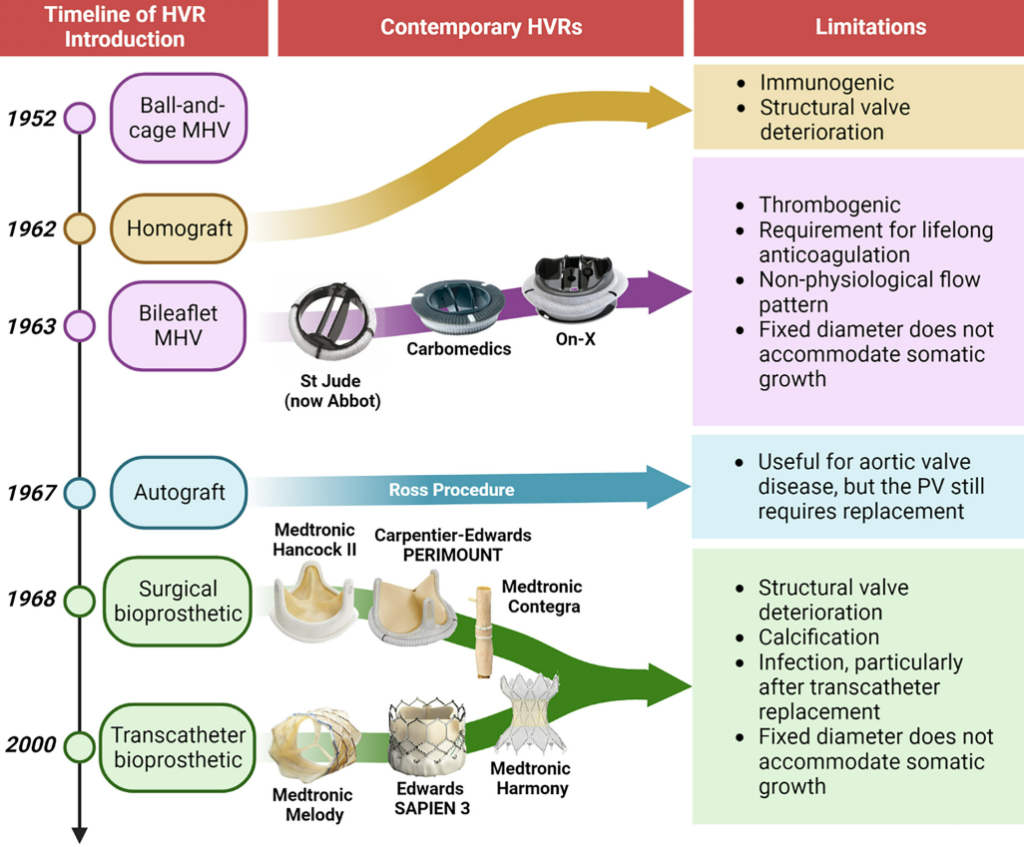
Pediatric pulmonary valve replacements: Clinical challenges and emerging technologies
Matthew Crago, David S. Winlaw, Syamak Farajikhah, Fariba Dehghani, Sina Naficy*
Congenital heart diseases (CHDs) frequently impact the right ventricular outflow tract, resulting in a significant incidence of pulmonary valve replacement in the pediatric population. While contemporary pediatric pulmonary valve replacements (PPVRs) allow satisfactory patient survival, their biocompatibility and durability remain suboptimal and repeat operations are commonplace, especially for very young patients. This places enormous physical, financial, and psychological burdens on patients and their parents, highlighting an urgent clinical need for better PPVRs. An important reason for the clinical failure of PPVRs is biofouling, which instigates various adverse biological responses such as thrombosis and infection, promoting research into various antifouling chemistries that may find utility in PPVR materials. Another significant contributor is the inevitability of somatic growth in pediatric patients, causing structural discrepancies between the patient and PPVR, stimulating the development of various growth-accommodating heart valve prototypes. This review offers an interdisciplinary perspective on these challenges by exploring clinical experiences, physiological understandings, and bioengineering technologies that may contribute to device development. It thus aims to provide an insight into the design requirements of next-generation PPVRs to advance clinical outcomes and promote patient quality of life.
Bioengineering and Translational Medicine 2023, e10501
https://doi.org/10.1002/btm2.10501
Figure. Introduction timeline of HVRs with application in the pediatric population. The first ball-and-cage mechanical valve was introduced in 1952, with the first generation of bileaflet mechanical valves introduced in 1963. The first homograft was implanted in 1962, the first use of an autograft in the Ross procedure was reported in 1967. The first chemically-treated bioprosthesis was described in 1968 and the first transcatheter bioprosthesis was successfully deployed in 2000.
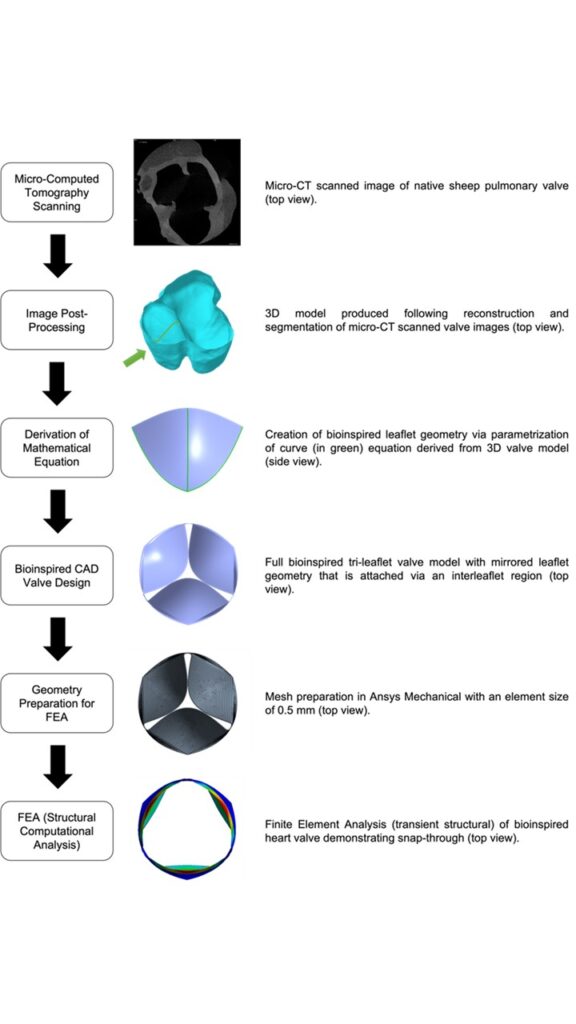
From Scan to Simulation—A Novel Workflow for Developing Bioinspired Heart Valves
Aeryne Lee, Syamak Farajikhah, Matthew Crago, Luke Mosse, David Frederick Fletcher, Fariba Dehghani, David Scott Winlaw, Sina Naficy*
Current heart valve replacements lack durability and prolonged performance, especially in pediatric patients. In part, these problems may be attributed to the materials chosen for these constructs, but another important contributing factor is the design of the valve, as this dictates hemodynamic performance and impacts leaflet stresses which may accelerate structural valve deterioration. Most current era bioprosthetic valves adhere to a fundamental design where flat leaflets are supported by commissural posts, secured to a sewing ring. This overall design strategy is effective, but functionality and durability can be improved by incorporating features of the native valve geometry. This paper presents a novel workflow for developing and analyzing bio-inspired valve designs computationally. The leaflet curvature was defined using a mathematical equation whose parameters were derived from the three-dimensional model of a native sheep pulmonary valve obtained via microcomputed tomography. Finite element analysis was used to screen the various valve designs proposed in this study by assessing the effect of leaflet thickness, Young’s modulus, and height/curvature on snap-through (where leaflets bend against their original curvature), geometric orifice area (GOA) and the stress in the leaflets. This workflow demonstrated benefits for valve designs with leaflet thicknesses between 0.1 and 0.3 mm, Young’s moduli less than 50 MPa, and elongated leaflets with higher curvatures. The proposed workflow brings substantial efficiency gains at the design stage, minimizing manufacturing and animal testing during iterative improvements, and offers a bridge between in vitro and more complex in silico studies in the future.
Journal of Biomechanical Engineering 2023, 145 (5), 055001
https://doi.org/10.1115/1.4056353
Figure: Process flow diagram of workflow from first stages of micro-CT scanning to computational analysis. The leaflet that is chosen to extract the geometry is highlighted by an arrow. A curve extraction tool was used to extract the geometry of the curve which is traveling along the length of the selected leaflet.
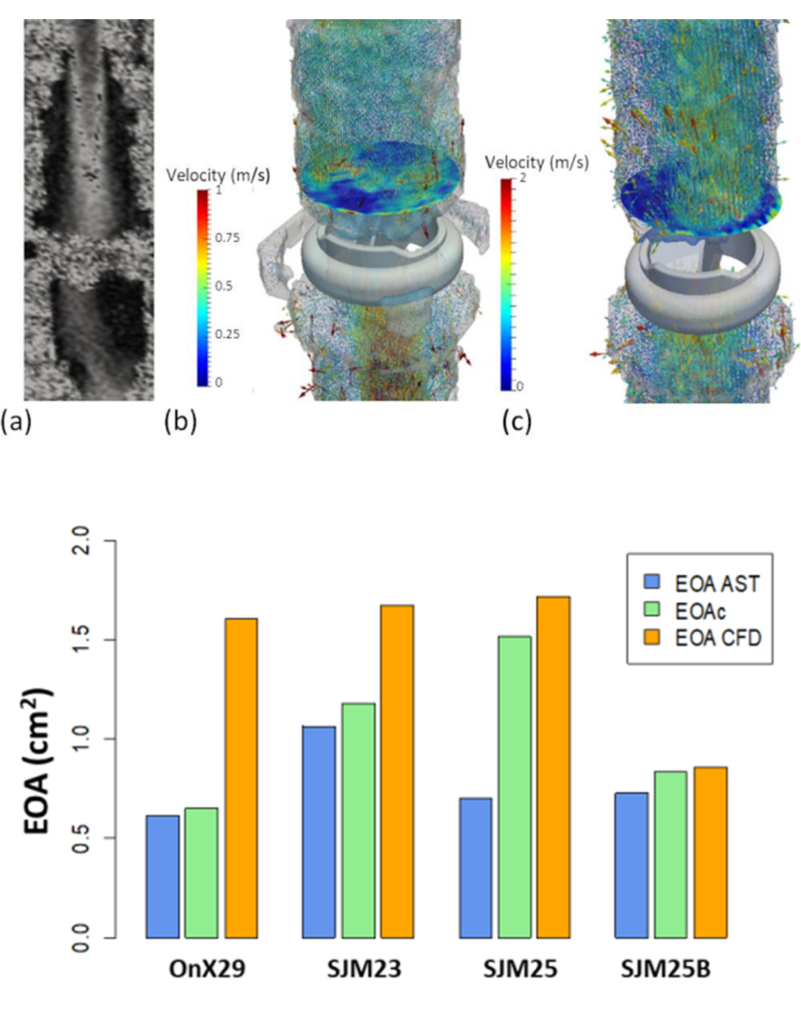
MRI Assessment of the Bi-Leaflet Mechanical Heart Valve: Investigating the EOA Using the Acoustic Source Term Method
Morgane Evin*, David Joannic, Aurélien Monnet, David F. Fletcher, Stuart M. Grieve, Jean-François Fontaine, Alain Lalande
This work aims at defining the Effective Orifice Area (EOA) derived from the acoustic source term (AST) method from 4D Phase-Contrast MRI data to provide a reference for the assessment of MRI valvular prostheses as part of a comprehensive cardiac exam. Three different Bileaflet Mechanical Heart Valves (BMHV) and a dysfunctional BMHV were tested invitro using 4D Phase-Contrast MRI and a numerical design of the experimental study was performed, including the influence of internal diameter, stroke volume, and heart rate. The EOA AST was computed based on the MRI 4D Phase-Contrast acquisition. EOAAST values vary by 15 mm from the BMHV centre and should be computed between 8 to 10 mm after the metallic “blurring” artefact (ranging from 18.9 to 23.4 mm from the BMHV). EOAAST values were found to be lower compared with numerical results in the appropriate plane. Detection of the BMHV dysfunction by continuity equation computed from 4D flow acquisition is limited based on EOAAST computations, while EOAc and velocities after the valves could more directly highlight a blocked leaflet. This multi-disciplinary study demonstrates the suitability of the EOA AST method to assess BMHV function using MRI. Translation to the clinic is feasible using an optimized 2D Phase-Contrast flow stack or a 4D Phase-Contrast minimal volume based on the recommendations provided.
Applied Sciences 2022, 12, 11771
https://doi.org/10.3390/app122211771
Figure: TOP) BMHV malfunction with MRI blurring around the BMHV (a), MRI 4D Phase-Contrast Acquisition with a superposed representation of normal functioning BMHV (b), and leaflet blocked MBHV (c) with AST values on the surface plane. BOTTOM) Comparisons of EOA for the four models of BMHV using MRI data (EOAAST (using the Acoustic Source term method)—blue and EOAC (using the continuity equation —green) and comparison with the numerical results EOA CFD (orange).
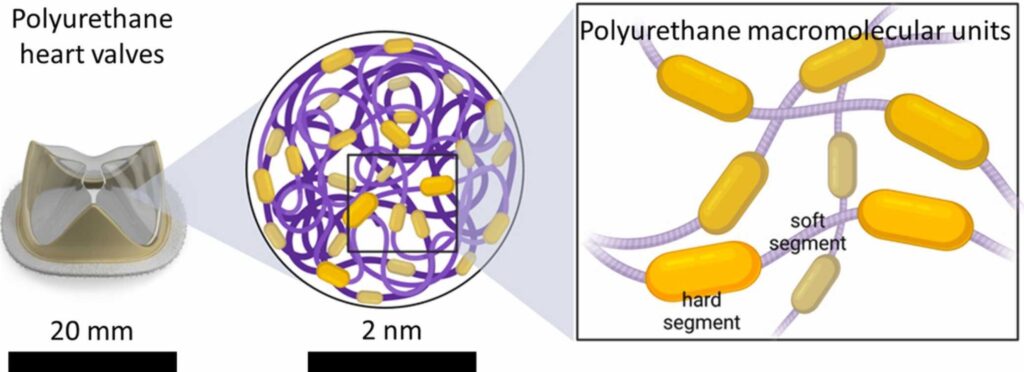
The evolution of polyurethane heart valve replacements: How chemistry translates to the clinic
Matthew Crago, Aeryne Lee, Syamak Farajikhah, Farshad Oveissi, David F. Fletcher, Fariba Dehghani, David S. Winlaw*, Sina Naficy*
Current heart valve replacements (HVRs) fail to comprehensively capture the physiological behavior of native heart valve tissues, allowing the burden of valvular heart disease to persist after surgical intervention. Through extensive research and clinical experience, it has become evident that material selection is central to the success of HVRs. One outstanding candidate material for next-generation HVRs is polyurethane. These polymers exhibit a unique segmented chemical structure that can imitate the mechanical strength and elasticity of soft biological tissues. Consequently, polyurethanes have long been investigated as HVR materials, however, progression to clinical utility has historically been impaired by the material’s susceptibility to biodegradation and calcification. This review presents a concise and critical analysis of past and recent investigations to elucidate the contemporary potential of polyurethane HVRs. Importantly, the overwhelming clinical failure of past prototypes necessitated a detailed examination to validate any further use of polyurethanes in HVR development. Ultimately, chemically modified polyurethanes along with different modifications have been introduced to tackle the shortcoming of the existing polyurethane HVRs and develop biostable biomaterials for next-generation HVRs.
Materials Today Communication 2022, available online
https://doi.org/10.1016/j.mtcomm.2022.104916
Figure: Schematic molecular structure of polyurethanes showing hard and soft segments of the polymer chains. At macroscopic scale, polyurethanes exhibit biocompatibility and mechanical properties which are suitable for heart valve replacement applications.
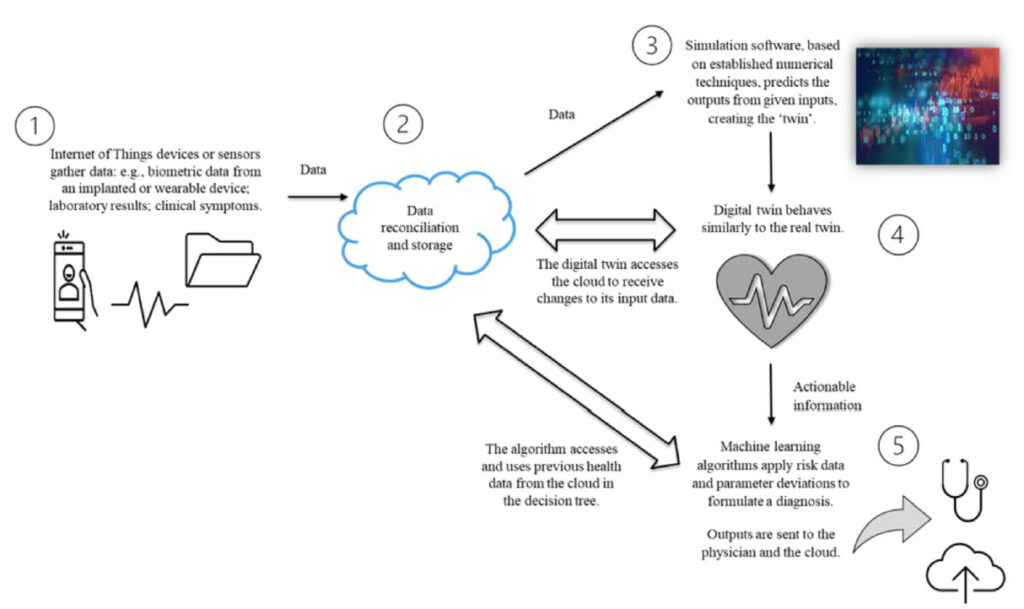
The Health Digital Twin to Tackle Cardiovascular Disease — A Review of an Emerging Interdisciplinary Field
Genevieve Coorey*, Gemma A. Figtree, David F. Fletcher, Victoria J. Snelson, Stephen T. Vernon, David Winlaw, Stuart M. Grieve, Alistair McEwan, Jean Yee Hwa Yang, Pierre Qian, Kieran O’Brien, Jessica Orchard, Jinman Kim, Sanjay Patel, Julie Redfern
Potential benefits of precision medicine in cardiovascular disease (CVD) include more accurate phenotyping of individual patients with the same condition or presentation, using multiple clinical, imaging, molecular and other variables to guide diagnosis and treatment. An approach to realising this potential is the digital twin concept, whereby a virtual representation of a patient is constructed and receives real-time updates of a range of data variables in order to predict disease and optimise treatment selection for the real-life patient. We explored the term digital twin, its defining concepts, the challenges as an emerging field, and potentially important applications in CVD. A mapping review was undertaken using a systematic search of peer-reviewed literature. Industry-based participants and patent applications were identified through web-based sources. Searches of Compendex, EMBASE, Medline, ProQuest and Scopus databases yielded 88 papers related to cardiovascular conditions (28%, n = 25), non-cardiovascular conditions (41%, n = 36), and general aspects of the health digital twin (31%, n = 27). Fifteen companies with a commercial interest in health digital twin or simulation modelling had products focused on CVD. The patent search identified 18 applications from 11 applicants, of which 73% were companies and 27% were universities. Three applicants had cardiac-related inventions. For CVD, digital twin research within industry and academia is recent, interdisciplinary, and established globally. Overall, the applications were numerical simulation models, although precursor models exist for the real-time cyber-physical system characteristic of a true digital twin. Implementation challenges include ethical constraints and clinical barriers to the adoption of decision tools derived from artificial intelligence systems.
NPJ Digital Medicine 2022, 5, 126
https://www.nature.com/articles/s41746-022-00640-7
Figure: Concepts in a digital twin model of the heart. A key aspect is the two-way data exchange between the digital model and the patient. The importance of big data and computational modelling in the process is evident.
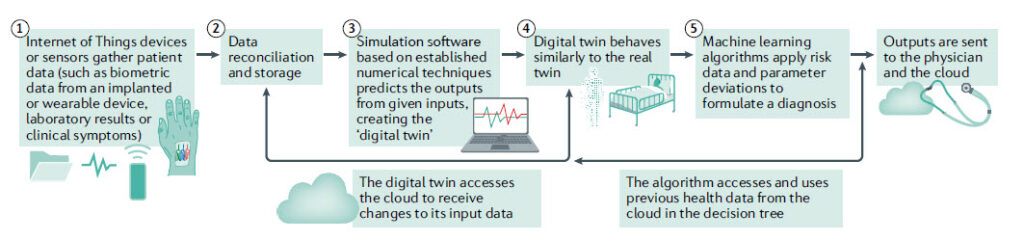
The Health Digital Twin to Tackle Cardiovascular Disease — A Review of an Emerging Interdisciplinary Field
Genevieve Coorey*, Gemma A. Figtree, David F. Fletcher, Victoria J. Snelson, Stephen T. Vernon, David Winlaw, Stuart M. Grieve, Alistair McEwan, Jean Yee Hwa Yang, Pierre Qian, Kieran O’Brien, Jessica Orchard, Jinman Kim, Sanjay Patel, Julie Redfern
Potential benefits of precision medicine in cardiovascular disease (CVD) include more accurate phenotyping of individual patients with the same condition or presentation, using multiple clinical, imaging, molecular and other variables to guide diagnosis and treatment. An approach to realising this potential is the digital twin concept, whereby a virtual representation of a patient is constructed and receives real-time updates of a range of data variables in order to predict disease and optimise treatment selection for the real-life patient. We explored the term digital twin, its defining concepts, the challenges as an emerging field, and potentially important applications in CVD. A mapping review was undertaken using a systematic search of peer-reviewed literature. Industry-based participants and patent applications were identified through web-based sources. Searches of Compendex, EMBASE, Medline, ProQuest and Scopus databases yielded 88 papers related to cardiovascular conditions (28%, n = 25), non-cardiovascular conditions (41%, n = 36), and general aspects of the health digital twin (31%, n = 27). Fifteen companies with a commercial interest in health digital twin or simulation modelling had products focused on CVD. The patent search identified 18 applications from 11 applicants, of which 73% were companies and 27% were universities. Three applicants had cardiac-related inventions. For CVD, digital twin research within industry and academia is recent, interdisciplinary, and established globally. Overall, the applications were numerical simulation models, although precursor models exist for the real-time cyber-physical system characteristic of a true digital twin. Implementation challenges include ethical constraints and clinical barriers to the adoption of decision tools derived from artificial intelligence systems.
NPJ Digital Medicine 2022, 5, 126
https://www.nature.com/articles/s41746-022-00640-7
Figure: Concepts in a digital twin model of the heart. A key aspect is the two-way data exchange between the digital model and the patient. The importance of big data and computational modelling in the process is evident.
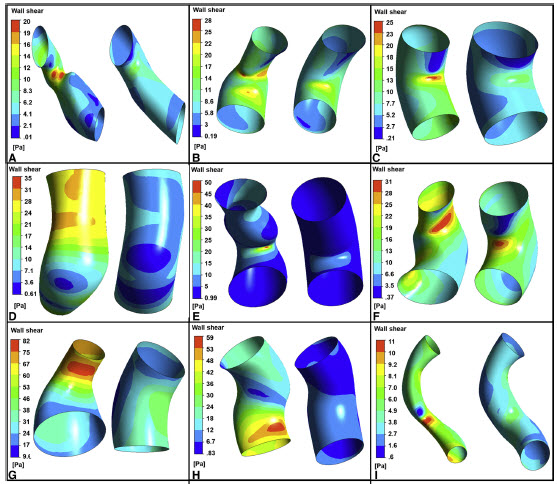
Evaluation of personalized right ventricle to pulmonary artery conduits using in silico design and computational analysis of flow
Pegah Ebrahimi, David Youssef, Gananjay Salve, Julian Ayer, Fariba Dehghani, David F. Fletcher, David S. Winlaw*
Right ventricle to pulmonary artery conduits are an essential part of many pediatric cardiac reconstructions. We demonstrate that in silico patient-specific design and analysis of the proposed conduit is feasible. Iterative customization improves hemodynamic performance with potential benefits in reducing right ventricle work and durability. This approach may be employed with new generation polymer-based conduits.
JTCVS Open 2020, 1(C), 33-48
https://doi.org/10.1016/j.xjon.2020.02.002
Figure: Comparison of the wall shear stress distribution between the initial (left) and modified geometries (right) at the beginning of the cardiac cycle for different patients. The reduction in wall shear stress and hence energy loss is evident.
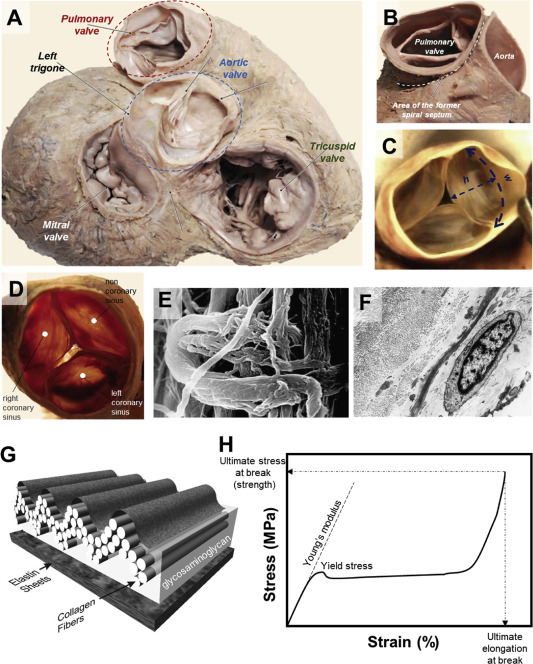
Materials and manufacturing perspectives in engineering heart valves: a review
Farshad Oveissi, Sina Naficy*, Aeryne Lee, David S Winlaw, Fariba Dehghani*
Valvular heart diseases (VHD) are a major health burden, affecting millions of people worldwide. The treatments for such diseases rely on medicine, valve repair, and artificial heart valves including mechanical and bioprosthetic valves. Yet, there are countless reports on possible alternatives noting long-term stability and biocompatibility issues and highlighting the need for fabrication of more durable and effective replacements. This review discusses the current and potential materials that can be used for developing such valves along with existing and developing fabrication methods. With this perspective, we quantitatively compare mechanical properties of various materials that are currently used or proposed for heart valves along with their fabrication processes to identify challenges we face in creating new materials and manufacturing techniques to better mimick the performance of native heart valves.
Materials Today Bio 2020, 5, 100038
https://www.sciencedirect.com/science/article/pii/S259000641930064X
Figure: Anatomy of human heart valves (A), the pulmonary valve in opened (B) and closed positions (C). Superior view of aortic heart valve (D) with SEM image of the aortic valve cusp in its connection point with fibrous ring (E), demonstrating collagen fibers wrapped circularly on collagen in the inner part of the fibrous ring. TEM image of the pulmonary valve cusp (F), representing collagen fibers in transverse and longitudinal direction, elastic fibers in transverse section, and fibroblasts. Schematic diagram of the multilayered configuration of an aortic valve cusp (G). A model tensile stress–strain curve showing various mechanical parameters referred to throughout this review (H).

Highly Porous, Biocompatible Tough Hydrogels, Processable via Gel Fiber Spinning and 3D Gel Printing
Sina Naficy*, Thi Yen Loan Le, Farshad Oveissi, Aeryne Lee, Jui Chien Hung, Steven G Wise, David S Winlaw, Fariba Dehghani
Conventional tough hydrogels offer enhanced mechanical properties and high toughness. Their application scope however is limited by their lack of processability. Here, a new porous tough hydrogel system is introduced which is processable via gel fiber spinning and 3D printing. The tough hydrogels are produced by rehydrating processable organogels developed by induced phase separation between two linear polymer chains capable of intermolecular hydrogen bonding. Through a slow sol–gel phase separation, highly porous gel networks made of hydrogen bonded polymer chains is formed. These organogels can be easily transformed to 3D printed multimaterial constructs or gel fibers, and after rehydration produce highly robust hydrogel structures. Although such hydrogels are highly porous and contain large amount of water, their strength can reach as high as 2000 kPa, with high elongation at break (≈900%), and tunable moduli ranging from 250 to 2000 kPa. The hydrogels have fracture energies larger than cartilage and demonstrate excellent load recovery because of their renewable hydrogen bond crosslinks. Furthermore, the hydrogels exhibit excellent hemocompatibility and in vitro biocompatibility. Such hydrogels can further expand the application of tough hydrogels and may serve as a model to explore the toughening mechanism of hydrogen bonded hybrid, tough hydrogel systems.
Advanced Materials Interfaces 2020, 7 (3), 1901770
https://onlinelibrary.wiley.com/doi/full/10.1002/admi.201901770
Figure: a) Gel processing of organogels b,c) 3D gel printing of a four-leaflet valve made of three components. d) 3D gel printing of the lower jaw with two gel components. The inset shows the 3D printing process. e–g) A multimaterial scaffold made of two gel components. The materials strongly fuse together so that no detachment occurs even under extreme deformation. h) A fully hydrated hydrogel fiber prepared via gel fiber spinning. Scale bars: 1.5 cm in (b)–(d), 1 cm in (e), 1.5 cm in (f), (g), and 1 cm in (h).
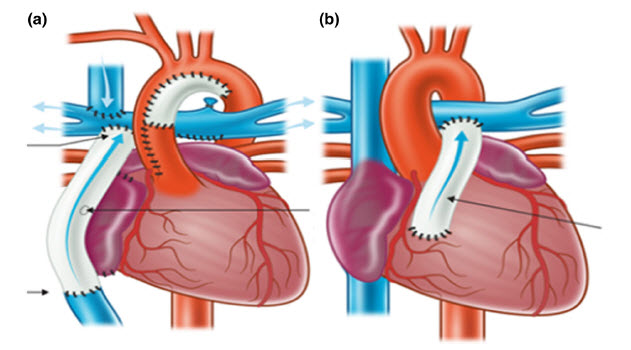
Current challenges and emergent technologies for manufacturing artificial right ventricle to pulmonary artery (RV-PA) cardiac conduits
Iman Manavithehrani, Pegah Ebrahimi, I. Yang, Sean Daly, Aaron Schindeler, A. Saxena, David G. Little, David F. Fletcher, Fariba Dehghani, David S. Winlaw*
Despite advances in modern surgery, congenital heart disease remains a medical challenge and major cause of infant mortality. Valved conduits are routinely used to surgically correct blood flow in hearts with congenital malformations by connecting the right ventricle to the pulmonary artery (RV-PA). This review explores the current range of RV-PA conduits and describes their strengths and disadvantages. Homografts and xenografts are currently the primary treatment modalities, however both graft types have limited biocompatibility and durability, and present a disease transmission risk. Structural deterioration of a replaced valve can lead to pulmonary valve stenosis and/or regurgitation. Moreover, as current RV-PA conduits are of a fixed size, multiple subsequent operations are required to upsize a valved conduit over a patient’s lifetime. We assess emerging biomaterials and tissue engineering techniques with a view to replicating the features of native tissues, including matching the durability and elasticity required for normal fluid flow dynamics. The benefits and limitations of incorporating cellular elements within the biomaterial are also discussed. Present review demonstrates that an alignment of medical and engineering disciplines will be ultimately required to produce a biocompatible and high-functioning artificial conduit.
Cardiovascular Engineering & Technology 2019, 10(2), 205-215
https://doi.org/10.1007/s13239-019-00406-5
Figure: Schematic of extracardiac Fontan (a) and RV-PA repair (b). Reused from The Royal Children’s Hospital Melbourne website.
